I’m Gilda Teissier, Executive Marketing Director at EvidentIQ and I recently met with Lise Radoszycki, COO at Carenity and Global Head of Data Science at EvidentIQ.
Over the past decade, we’ve observed a rise in patients’ power and engagement regarding their own care.
The concept of Evidence-based medicine has also emerged with the goal of delivering the right care at the right time to the right patient. One of the core concepts of Evidence-based medicine is patient preference.
Industry, regulators, HTA bodies and players are showing more and more interest in the use of patient preference studies. The goal is to help incorporate the patient perspective into clinical drug development, care management, and healthcare decision-making.
In this context, most of the pharmaceutical companies want to be patient-centric, but it’s not always clear how and when to include patient perspective into the product development cycle.
The following interview with Lise Radoszycki, COO at Carenity and Global Head of Data Science at EvidentIQ, will give us an overview of preference study methodologies focusing on Discrete Choice Experiments (DCE), and will share light on how to optimize DCE design to improve the quality on patient studies.
Gilda: First of all, could you explain to us what a preference study is?
Lise: Of course, patient preference studies are based on patient preference information, that reflects what treatment attributes matter to patients, how much these matter to them and how they make trade-offs between treatments attributes.
Describing trade-offs allows us to evaluate compromises done by patients when they select their preferred treatment option. To illustrate this concept, I will ask you 3 questions:
1. Do you prefer to be rich or to be poor? I guess that your answer will be rich. In the same way, if I ask you, do you prefer to be sick or to be healthy? Again, the answer is easy, you will mostly say “healthy.”
2. Now, if I ask you “Do you prefer to be rich and sick or poor and healthy?” The choice is more complicated, and you will need to make some compromises or trade-off.
For each of these 3 types of information, qualitative or quantitative methodologies exist. Qualitative methods are mainly based on focus groups or semi-structured interviews, whereas quantitative methods are mainly based on questionnaires administered directly to patients.
Among the quantitative methods there are two main types of methodologies: the revealed preference methods and the stated preference methods.
Unlike revealed preference techniques that use observations on real choices made by respondents, stated preference methods ask patients how much they value something. This type of methodology allows to evaluate a treatment that is not yet on the market or a drug that targets a restricted population of patients such as a rare disease or a specific disease subtype. Stated preference methods are widely used in healthcare. In this type of method, the subject is asked how much they value something and here is where the Discrete Choice Experiment comes in.
Gilda: What is a Discrete Choice Experiment?
Lise: A DCE is a stated preference method increasingly used in healthcare to elicit patient preferences. In a DCE, participants are asked to state their preferred choice between several competing treatment alternatives. In general, there are two treatment options. The features of these treatments are called attributes. Each attribute is decomposed into levels. Each treatment alternative presented to participants is a combination of treatment attributes and levels. The question asked to patients might be: “If you were offered these two treatments, which one would you choose?”
“In a DCE, participants are asked to state their preferred choice between several competing treatment alternatives.”
Gilda: And which would be the main questions that need to be asked in order to develop a successful DCE?
Lise: For me there are three questions that help create the best study design. You can use these best practices for all patient studies, not just preference studies.
The first question is: Which research question is best suited for the strategic objectives to be achieved by measuring patient preferences?
The second: Which study population would be most appropriate to answer the defined research question?
Finally: What is the most appropriate methodology to answer the research question while considering the operational constraints?
Gilda: If we take this into consideration, it would mean that the DCE is not the right method for every preference study?
Lise: Even if the DCE is considered as a gold standard, it is not always the most appropriate method. Indeed, it is important to highlight that the main guidelines on preference studies published by the NICE, the FDA or ISPOR do not impose a particular methodology, but propose criteria for assessing the quality of patient studies.
For example, if the goal is to evaluate preferences regarding several topics (Qol, care pathway) the DCE is not the most appropriate methodology. Same if you want to evaluate the perception of a specific treatment option testing some pre-defined hypothesis. It is also very important to consider the operational constraints such as the prevalence of the study population, the timelines, the budget allocated to the study, etc.
Gilda: There are 5 key steps to a preference study with DCE methodology, could you tell us more about them?
Lise: Briefly, we could say that the first step is to identify the attributes of the treatment. The second, is to identify and define the levels of each attribute with methods such as literature research, and qualitative interviews. The third step is to create scenarios and choice sets with an experimental design method, which will be used to select a reduced sample of choices (named “fractional factorial” design). The fourth step is Statistical Modeling based on the random utility model. And the final key to a DCE study is the interpretation of results, where it is crucial to identify those that have a negative or positive impact on the preference of a treatment. Patient preference studies generate insightful data that can be used at each step of the medical product life cycle.
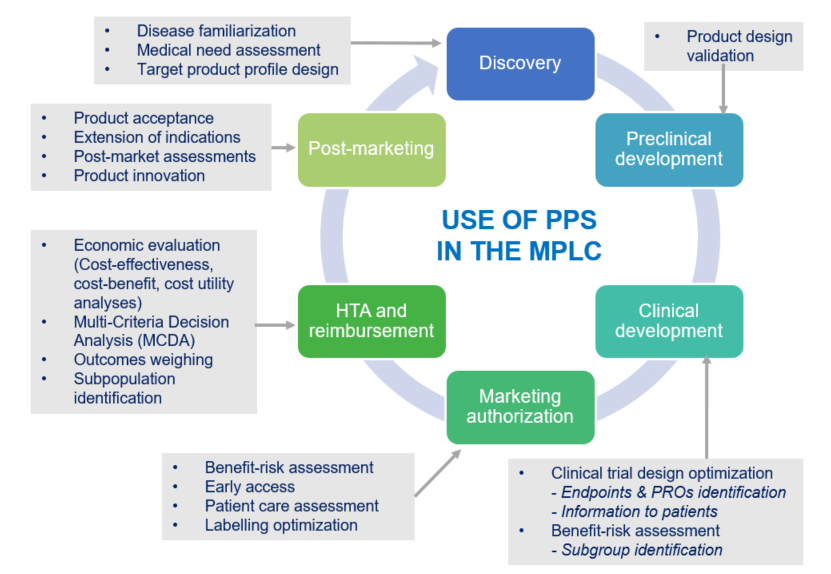
Gilda: Do patient preference studies really make a difference in regulatory decision-making?
Lise: This is a very interesting question. Several Authorities (such as NICE or FDA) invite companies to start a conversation with them about using patient preference information to support their submission. Over the past years, several guidelines and initiatives have been developed to implement and improve quality standards (such as PREFER or IMI).
There is therefore, a real desire to include patient studies in regulatory decision-making. Nevertheless, the level of consideration given to this type of study is very variable and highly depends on the quality of the study.
For example, Janssen included in its dossier a preference study to support Esketamine, a treatment for resistant depression. The goal of this DCE study was to assess patient perspective on benefit-risk trade-offs. In their HTA dossier, outcomes importance was reinforced by the results of the patient study. The advisory committee of the FDA said that it was taken into account in their decision, but we do not know how much influence it really had at the end.
Gilda: Before leaving, could you give us some final tips to a successful DCE study?
Lise: First, it is really important to make sure your participants fully understand the study materials to reduce uncertainty caused by health numeracy/literacy.
For this purpose, you can use a pilot study or a cognitive debriefing to pre-test the questionnaire and the material associated with the study with a limited number of patients.
The second advice would be to ensure representativeness of the sample and generalizability of results.
Another important thing to remember is that in order to improve the quality of the results, it is crucial to minimize the potential cognitive biases, such as framing bias, anchoring bias, ordering or labelling effects or simplified heuristics.
The final advice that I can give is to think carefully about the statistical analysis plan and data management plan in order to ensure logical soundness and robustness of data analysis.
Gilda: Thank you for your time.
A webinar addressing this same topic was recently conducted by Carenity and Takeda through the Xtalks platform. If you want to watch the replay you can access it by clicking here: ACCESS WEBINAR
For more information about EvidentIQ’s and Carenity’s solutions click here.

